Delta-8 THC Ban: What Brands Must Do Now
Last Updated: March 26, 2026
If your company still sells Delta-8 vape products, the core problem in 2026 is not abstract legal theory. It is operational panic: what happens to your inventory, your interstate shipments, your retail relationships, and your next product line once the federal rule change takes effect on November 12, 2026? The answer is uncomfortable but clear. Public Law 119-37, signed on November 12, 2025, rewrites the federal hemp framework in a way that leaves most legacy Delta-8 vapes with no realistic path to continued federal hemp compliance.
This article is designed to solve three urgent questions at once. First, how serious is the Delta-8 THC ban in practical terms? Second, what does compliance actually require before November 12, 2026? Third, how can brands survive without freezing up, dumping inventory blindly, or chasing fake loopholes? For business education only, not legal advice, the short answer is this: panic is understandable, but waiting is the most dangerous option.
Why the Delta-8 THC Ban Feels Like an Overnight Shutdown
The federal trigger is already in place. Section 781 of Public Law 119-37 takes effect on November 12, 2026, and it does two things that matter immediately to Delta-8 operators. It excludes many intoxicating hemp-derived cannabinoid products from the federal hemp definition if they contain more than 0.4 milligrams combined total per container of THC and similarly acting cannabinoids, and it also excludes cannabinoids that were synthesized or manufactured outside the plant.
That is why the new rule feels like a master switch. A standard 1 g Delta-8 cartridge is not slightly over the new limit. It is usually hundreds or thousands of times over it. If a cart holds around 800 mg of Delta-8, a 0.4 mg cap is not a reformulation problem. It is a category-collapse problem. For most brands, that means the existing Delta-8 THC ban conversation is no longer about lobbying narratives or semantic packaging. It is about how quickly they can stop relying on SKUs that will not fit the post-November federal definition.
Congressional Research Service further notes that products falling outside the amended hemp definition may instead be treated as marijuana or as CSA-regulated THC under federal law. That is the sentence behind the panic. Not every business will face enforcement the same way, but every business that treats late-2026 interstate Delta-8 trade like ordinary hemp commerce is taking a materially higher legal risk than before.
-
- What brands should do now: freeze expansion plans for new Delta-8 vape SKUs tied to 2026 holiday sales.
-
- What brands should do in the next 30 days: separate inventory into sell-through, reformulate, repurpose, and legal-review categories.
-
- What brands should do before Q3 2026 ends: align hardware, packaging, and distribution around a non-Delta-8 survival model.
Why the Old “0.3% Delta-9” Defense No Longer Calms Anyone Down
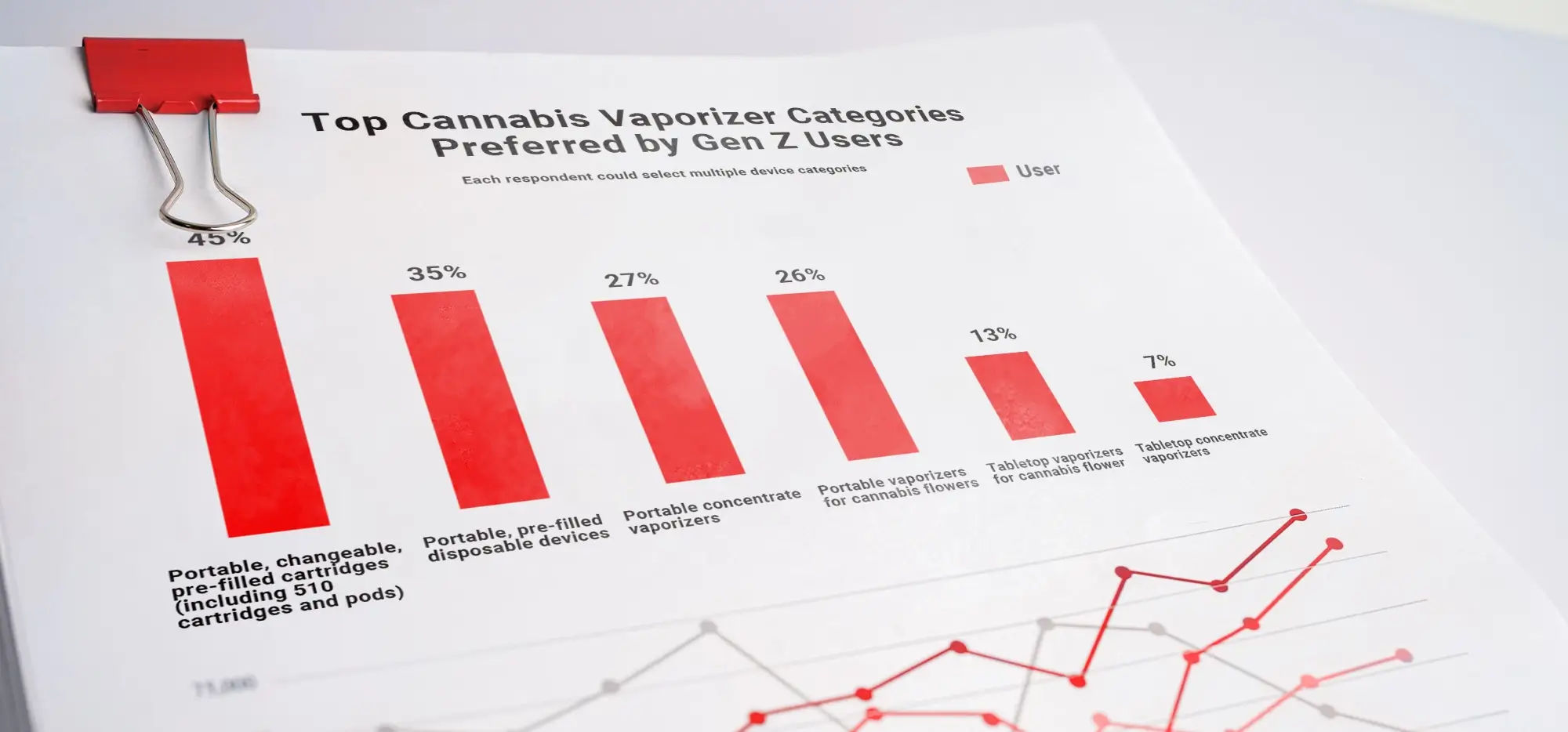
For years, many brands built their Delta-8 business on one key sentence from the 2018 Farm Bill era: if the material stayed under 0.3% Delta-9 THC on a dry-weight basis, it could be sold as hemp. That logic always had limits, but in 2026 it becomes dramatically less useful. The new federal framework explicitly brings THCA into the analysis and uses a more expansive approach to total THC and similarly acting cannabinoids in finished products.
For technical audits, operators still often use the familiar THC conversion shorthand:
Total THC = (THCA * 0.877) + Delta-9 THC + Delta-8 THC + …
That formula reflects USDA-style total THC logic for THCA conversion. But the larger business lesson is more important than the chemistry: labels that once sounded safe, such as “hemp-derived” or “under 0.3% Delta-9,” no longer reassure buyers, regulators, or serious retail partners when the finished product is clearly intoxicating.
This also explains why synthetic conversion and isomerization routes are under heavier pressure. FDA warns that concentrated Delta-8 is often made from hemp-derived CBD through chemical conversion and that the process may involve harmful by-products or unsafe contaminants. The 2026 federal amendment then makes that manufacturing history legally relevant by excluding certain cannabinoids synthesized or manufactured outside the plant from covered hemp definitions. That closes off the emotional comfort many operators used to get from technical wording alone.

State-Level Precedents: Why California Already Showed the Future While Texas and Florida Keep Brands Guessing
If the federal change creates panic, the state map explains why many operators felt pressure long before November 2026. California’s AB 8, signed on October 2, 2025 and effective January 1, 2026, sharply tightened the market by banning THC-containing hemp inhalables from in-state sale and imposing strict standards on hemp ingredients in foods and supplements. For everyday business operators, that means the gas-station and convenience-store Delta-8 era was already collapsing in one of the country’s most important markets.
Texas and Florida remain more complicated, which is why they are so emotionally confusing for brands. Texas State Law Library says Delta-8 remains legally unclear because a temporary injunction still blocks the state’s attempted scheduling change while litigation continues. Florida, meanwhile, still operates under a testing, labeling, and age-restriction framework for hemp extract, including a 21+ requirement for inhalable hemp extract, rather than an express statewide Delta-8 ban in the statute itself.
The problem is that patchwork rules can create false emotional relief. A brand may still have short-term room in one state, but that does not undo the federal November 12, 2026 deadline. State ambiguity is not a substitute for a federal survival plan.
| Jurisdiction | 2026 posture | Practical business takeaway |
|---|---|---|
| Federal | Rule enacted, effective November 12, 2026 | Most Delta-8 vape SKUs lose a viable federal hemp path |
| California | AB 8 in effect from January 1, 2026 | THC hemp inhalables are already removed from ordinary retail sale channels |
| Texas | Litigation and injunction still shape access | Temporary room does not equal long-term certainty |
| Florida | Regulated hemp-extract framework remains | Testing and age-gates matter now, but federal exposure still rises in November |
| Colorado | Converted-cannabinoid enforcement stance | CBD-to-Delta-8 style conversion was already under official pressure |
Industry Growing Pains and Transformation: 3 Survival Paths in the Post-Delta-8 Era
1. Migrating to THCA: Commercially Tempting, but No Longer a True Federal Escape Hatch
Many operators have tried to treat THCA as the clean replacement for Delta-8 because it sounds more plant-native and less conversion-dependent. But 2026 limits that story. Federal hemp-testing logic already counts THCA through the 0.877 conversion factor, and the amended federal framework explicitly brings THCA into the broader total-THC analysis. So THCA may be commercially relevant in some channels, but it is not a universal legal safe haven.
That said, the migration trend still changes hardware demand. Brands moving toward pre-rolls, dry-herb devices, or regulated live rosin formats need different heating profiles, different clog-management strategies, and different customer education. The winners here are not the brands with the loudest loophole story. They are the brands that retool their product system faster than competitors do.
2. The Medicalization of High-Purity CBD and Minor Cannabinoids
The second survival path is more defensive but often more durable. Instead of trying to preserve an intoxicating hemp identity at all costs, some brands are moving toward cleaner non-intoxicating portfolios built around CBD, CBG, CBN, and stronger documentation. This works best when brands stop making vague wellness hype the center of the sale and start using pharmacy-style discipline: better testing, cleaner ingredient narratives, more cautious marketing language, and clearer device compatibility.
That strategy matters because much of the regulatory hostility around Delta-8 is tied to fear of residues, conversion chemistry, and poorly controlled retail exposure. FDA warns about adverse events, poison-center calls, appealing packaging, and contaminants created during manufacturing. A brand that can visibly distance itself from that risk profile may lose the impulse-buy audience, but it can gain retailer trust and longer-term channel stability.
3. Breaking Barriers: From Hemp Brand to Licensed Adult-Use Operator
The third path is the boldest and usually the most expensive. Some well-funded brands will decide that if the Delta-8 THC ban wipes out the hemp-intoxication model, the smart move is to enter licensed cannabis markets directly. That requires a very different operating system: state licenses, lab testing, tax planning, compliant distribution, and far stricter packaging and traceability.
Still, this path can preserve what actually made some hemp brands strong in the first place. Speed to market, branding, product storytelling, and hardware innovation all still matter in licensed cannabis. What changes is the level of legal structure around them. For some companies, the real survival move is not saving the old model. It is graduating out of it.
How to Reduce Operational Pain While You Rebuild the Portfolio
Compliance is not just a legal memo. It becomes physical the moment a brand changes oil viscosity, cannabinoid profile, or intended market. Crystallization-prone oils, purified formulations, and terpene-heavy extracts all place different stress on a device than classic Delta-8 distillate did. Artrix’s Lilmon Vision is a useful example because it offers adjustable voltage, preheat-duration control, and compatibility positioning across distillates, live resin, rosin, and related formats. For brands dealing with clogging anxiety, that kind of controlled warm-up matters.
Leak prevention also becomes more important as brands move into thinner or more formulation-sensitive compliant oils. Cubox uses a postless architecture, dual-air-channel design, and flat ceramic heating, while Lilcube uses a postless structure and dual airflow aimed at stable performance and clog reduction for terpene-rich oils. The practical takeaway is simple: legal reformulation without hardware reformulation often creates a second failure point.
There is also a survival question hidden inside old inventory. Some hardware assets from the Delta-8 era may still be reusable if the voltage range, preheat behavior, and airflow can be tuned for licensed distillate, live resin, or other compliant products. That will not rescue non-compliant Delta-8 inventory, but it can reduce the cost of the transition and calm some of the panic around stranded hardware.
FAQ: What Operators Are Really Asking When They Panic About Delta-8 Inventory
Q: Can I keep shipping Delta-8 inventory across state lines after November 12, 2026?
That becomes far riskier. CRS explains that products falling outside the amended hemp definition may instead be treated as marijuana or as controlled THC under federal law. In practical terms, interstate shipment after the deadline should not be treated like ordinary hemp commerce without legal review.
Q: Should I panic-sell every Delta-8 SKU right now?
Not blindly. A smarter approach is inventory triage: identify what can be sold lawfully before the deadline, what should be stopped immediately due to channel or state risk, and what should be reviewed for reformulation, write-down, or disposal planning. Panic is understandable, but undisciplined discounting can create its own regulatory and reputational damage.
Q: What does the 0.4 mg cap really mean for a 1 g cart?
It means most standard Delta-8 carts are nowhere near compliance. The federal cap applies per container, not as a loose marketing concept. A cart holding hundreds of milligrams of Delta-8 is not close to the threshold. It is overwhelmingly above it.
Q: Are there any new loopholes left?
Not clean ones. THCA is also implicated by total-THC logic, converted-cannabinoid models are under heavier scrutiny, and state restrictions continue to narrow room for improvisation. In 2026, the serious business strategy is adaptation, not loophole hunting.
Conclusion: The Way to Survive the Delta-8 THC Ban Is to Move Before You Feel Ready
The real danger of the 2026 Delta-8 THC ban is not only legal exposure. It is paralysis. Brands that stay stuck between denial and panic may lose the time they still have to manage inventory, repair retailer relationships, redesign hardware strategy, and choose a post-Delta-8 business model. The market is not waiting for everyone to feel emotionally ready.
The brands most likely to survive are the ones that act early, document aggressively, and stop confusing temporary ambiguity with long-term safety. In a policy environment this unstable, compliant hardware, formulation discipline, and transparent quality systems become your last reliable line of defense. If your team needs to rethink device compatibility, repurpose legacy hardware, or build a more resilient vape portfolio for the 2026 regulatory era, contact Artrix for hardware solutions built around the realities of the post-Delta-8 market.

















-1.webp)
-1.webp)
-2.webp)